Abstract
Introduction
Since the commercial approval of chimeric antigen receptor T cell (CAR-T) therapies, administration and toxicity monitoring have largely been in an inpatient setting due to the risk of significant toxicities such as cytokine release syndrome (CRS) and neurotoxicity in the first 30 days. Administration in the outpatient setting can be safe and cost-effective. Here we report the cost savings and adverse events of CAR-T in an outpatient setting as compared to the inpatient setting.
Methods
Cost differences of the commercial CD19 CAR-T axicabtagene ciloleucel (axi-cel) and tisangenlecleucel (tisa-cel) among inpatient and outpatient settings in 2020 were investigated using the Vizient Database®. Cost analysis for both settings was done for the initial 30 days post-CAR-T infusion. There were no billing codes for CRS and neurotoxicity till 2021. Clinical surrogates such as fever, hypotension, hypoxia, sepsis were used for CRS while febrile convulsion not otherwise specified (NOS), febrile seizure NOS, altered mental status, somnolence, stupor, and coma were used for neurotoxicity. ICD 10 codes for adverse effects were also used.
Results
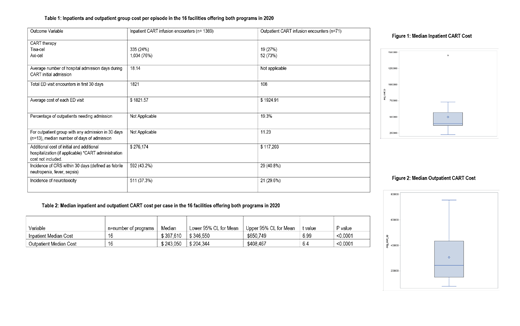
In 2017-2020, there were 81 organizations in the database that performed CAR-T procedures. In 2020, there were 1369 inpatient and 71 outpatient encounters, which were analyzed for cost and adverse events. (Table 1) The incidence of CRS was 43.2% (n=592) and 40.8% (n=29) in inpatient and outpatient groups, while that of neurotoxicity was 37.3% (n=511) and 29.6% (n=21) respectively. For cost analysis, we included the 16 centers (22% of all centers) that offered both inpatient and outpatient administration in 2020. Median inpatient cost was $397,610 ($346,550-$650,749) and median outpatient group cost was $243,050 ($204,344-$408,467). An analysis of variance (ANOVA) was run between inpatient and outpatient cases was found to be significant (P<0.0001). (Table 2) (Figure 1) (Figure 2)
Conclusion
As the field of CAR-T therapy continues to grow, outpatient programs are likely to increase. Incidence of adverse effects was lower in the outpatient group, likely patient selection effect. This data suggests that outpatient CAR-T therapy is feasible cost-effective and has the potential to grow and improve value. While it appears to be an attractive option, there is a need for more studies on patient selection and creating a robust outpatient infrastructure is needed.
Mahmoudjafari: Incyte: Membership on an entity's Board of Directors or advisory committees; Omeros: Membership on an entity's Board of Directors or advisory committees; GSK: Membership on an entity's Board of Directors or advisory committees. McGuirk: EcoR1 Capital: Consultancy; Novartis: Research Funding; Fresenius Biotech: Research Funding; Novartis: Research Funding; Astelllas Pharma: Research Funding; Bellicum Pharmaceuticals: Research Funding; Kite/ Gilead: Consultancy, Honoraria, Other: travel accommodations, expense, Kite a Gilead company, Research Funding, Speakers Bureau; Gamida Cell: Research Funding; Magenta Therapeutics: Consultancy, Honoraria, Research Funding; Juno Therapeutics: Consultancy, Honoraria, Research Funding; Allovir: Consultancy, Honoraria, Research Funding; Pluristem Therapeutics: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal